Absolute Entropy Table. The symbol for entropy is S and the standard entropy of a substance is given by the symbol S o indicating that the standard entropy is determined under standard conditions. When the noble gas gets heavier this induces more entropy.
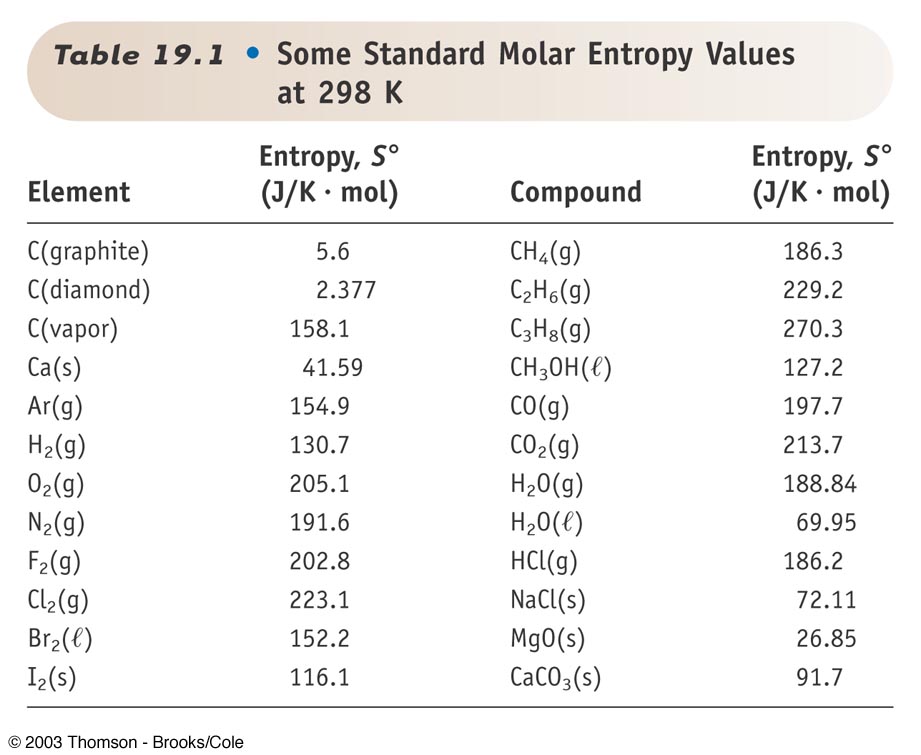
The change in standard absolute entropy ΔS for this reaction is. There are some clear trends. Useful way of measuring entropy is by the following equation.
The concept entropy was first developed by German physicist Rudolf Clausius in the mid-nineteenth century as a thermodynamic property that predicts that certain spontaneous processes are irreversible or impossible.
Are there tables of absolute as opposed to standard molar entropies of water at various temperatures. 100 rows Standard Heats and Free Energies of Formation and Absolute Entropies of Elements. I wanted to calculate the entropy of the data in a table. When water molecules escape from the liquid phase and enter the gas phase the entropy of this system increases by 1188 J K-1 mol-1.
